Policy dividends released, medical silk products require more 'ID cards'
01 Ten support measures, high-end medical device industry welcomes innovation turning point
With the continuous development of modern medical technology, medical devices have been widely used for disease diagnosis, prediction, monitoring, treatment or relief. In July of this year, the National Medical Products Administration issued the Announcement on Measures to Optimize Full Life Cycle Supervision and Support the Innovation and Development of High end Medical Devices (No. 63 of 2025), which undoubtedly injected strong impetus into the development of the high-end medical device industry.
The announcement clearly states that we will fully support major innovations in high-end medical devices, promote the application of more new technologies, materials, processes, and methods in the medical and health field, especially in key areas such as medical robots, high-end medical imaging equipment, artificial intelligence medical devices, and new biomaterials medical devices that shape the new quality and productivity of medical devices. We also propose ten support measures, including optimizing special approval procedures, to provide clear policy guidance for the innovative development of the industry.
02 Strict control, medical device registration becomes the "gatekeeper" of innovation
The development of medical device technology and industry is closely related to national health security. The independent innovation and localization of high-end medical devices are the technical support and important guarantee for the healthy development of China's medical system. However, currently most high-end medical devices in clinical practice still rely on imports, and this situation urgently needs to be broken through.
In recent years, the scale of China's medical device market has shown a sustained growth trend, driven mainly by three aspects: firstly, the acceleration of population aging and the rising incidence of chronic diseases; Secondly, the public's awareness of health has been enhanced, and the demand for medical care continues to be released; Thirdly, the national policy continues to intensify, supporting innovation and breakthroughs in high-end medical devices.
At the same time, the safety and effectiveness of medical devices acting on the human body directly affect human health and life safety, and must be strictly controlled. Therefore, medical device registration has become a key link in product launch, and its scientific and standardized nature is particularly important.
03 Registration Certificate=Market ID Card+Technical Passport?
Medical device registration refers to the process in which the food and drug regulatory authorities, based on the application of the applicant for medical device registration, conduct a systematic evaluation of the safety and effectiveness research and its results of the medical devices intended for marketing in accordance with legal procedures, in order to decide whether to approve their application. Medical device registration is divided into domestic and overseas, and is classified into three categories based on their safety and risk levels:
The first type of medical device has a low level of risk, and implementing routine management can ensure its safety and effectiveness.
The second type is medical devices with moderate risk that require strict control and management to ensure their safety and effectiveness.
The third category is medical devices with higher risks that require strict control and management to ensure their safety and effectiveness.
For enterprises, the medical device registration certificate has a dual significance: on the one hand, it is the "ID card" for the legal entry of enterprise products into the market. Only by obtaining the registration certificate can enterprises promote their products to the market for sale; On the other hand, the registration certificate is an authoritative certification of product safety and effectiveness, especially in the field of high-end medical devices. Obtaining the registration certificate represents that the technical strength of the enterprise has been strictly verified by the national regulatory authorities. This certification is not only related to market access, but also an important manifestation of a company's core competitiveness.
04 With the addition of the Double Three Class Certificate, Xingyue Biotechnology has achieved two major breakthroughs in the field of new biomaterials
In the field of new biomaterials and medical devices, the Xingyue Biotechnology team was the first to explore the medical silk track as early as 2005. As a natural polymer material in biomaterials, silk fibroin is harmless to the human body, safe and reliable, and has good biocompatibility. It is suitable for developing functional materials and can be prepared into materials with different properties such as fibers, films, sponges, powders, etc. through research and development technology.
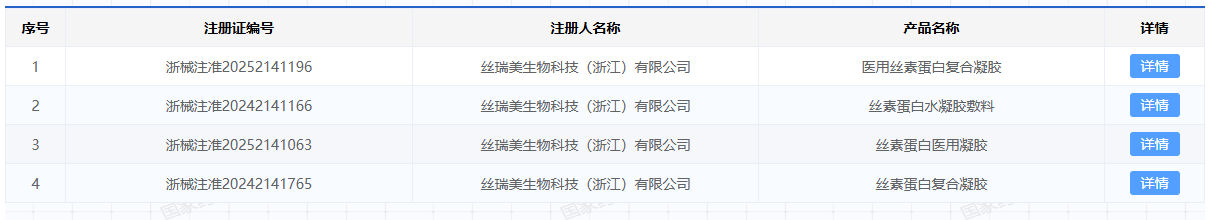
At present, after nearly 20 years of technological accumulation, Xingyue Biotechnology has successfully applied silk fibroin protein to fields such as medical devices, medical beauty, and functional skincare. In the field of medical devices, Xingyue Biotechnology and its subsidiary Siromei Biotechnology have accumulated 10 medical device registration certificates, of which 2 are Class III medical device registration certificates.
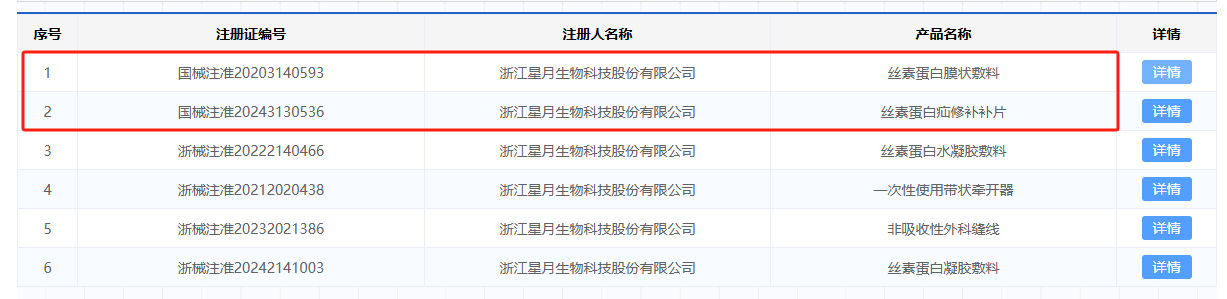
The silk fibroin membrane dressing obtained a Class III medical device registration certificate in 2020 and is a transparent regenerated silk fibroin membrane. This product is a transparent, dense, smooth, and breathable film made from 5A grade or higher mulberry silk, suitable for covering the skin supply area.
The silk material hernia repair product, silk fibroin hernia repair patch, which has been clinically validated by multiple centers, obtained a Class III medical device registration certificate in 2024 and was included in the "Zhejiang Province Key New Material First Batch Application Demonstration Guidance Catalog (2024 Edition)". This product is a mesh patch made of mulberry silk through processes such as warp knitting and degumming. Its composition is silk fibroin and it is suitable for open abdominal external repair of inguinal hernia.

With the implementation of the National Medical Products Administration's optimized full lifecycle regulatory policies, the high-end medical device industry is facing unprecedented development opportunities. With its forward-looking layout in the field of new biomaterials and nearly 20 years of technological accumulation, Xingyue Biotechnology will further promote the leap from basic research to industrial application.









