7 main document registrations, leveraging the main document system of Xingyue Biology to open up the application pathway of silk fibroin protein
01 What is the Medical Device Master Document System
The main document system for medical devices is an important technical document submission mechanism in medical device regulation. This information is submitted by its owner to the medical device regulatory agency as a confidential document containing key technical parameters, which is used to authorize medical device product applicants to use it when applying for medical device registration and other matters.
02 Differentiation in the development of the main document system both domestically and internationally
Throughout foreign regulatory practices, the main document system for medical devices has shown a differentiated development trend. The Center for Devices and Radiological Health (CDRH) of the US FDA established relevant management regulations as early as 1997, the Japanese Ministry of Health, Labour and Welfare issued relevant master document notices for specific polymer materials in 2000, and the European Union recorded relevant requirements for drug master documents in its directives. Since the National Medical Products Administration of China launched relevant research in 2018, it has gradually established a medical device master document registration system that is in line with the national conditions. In 2021, it issued the "Announcement on the Registration Matters of Medical Device Master Documents".
03 The number of silk fibroin master document registrations has surged
Medical devices play an irreplaceable role in the prevention, diagnosis, treatment, and rehabilitation of diseases, and their safety and effectiveness are directly related to the life and health of patients. The performance of medical devices largely depends on the raw materials used, so the selection of raw materials is crucial. The common raw materials for medical devices currently include metal materials (such as titanium alloys, stainless steel), polymer materials (such as polyethylene, polyurethane), ceramic materials (such as zirconia), etc.
In recent years, with the deepening of research on biomaterials, silk fibroin has emerged as a new type of raw material. This natural protein extracted from silk has excellent biocompatibility, degradability, and mechanical properties, demonstrating unique advantages in fields such as wound repair, tissue engineering scaffolds, and drug delivery systems.
With the deepening of research on the application of silk fibroin in the field of medical devices, the number of silk fibroin master document registrations has also significantly increased. According to data from the website of the Medical Device Technical Evaluation Center of the National Medical Products Administration, as of May this year, there have been 39 registered silk fibroin master documents, involving 11 innovative enterprises. This development trend fully confirms the broad application prospects of silk fibroin materials in the field of high-end medical devices.
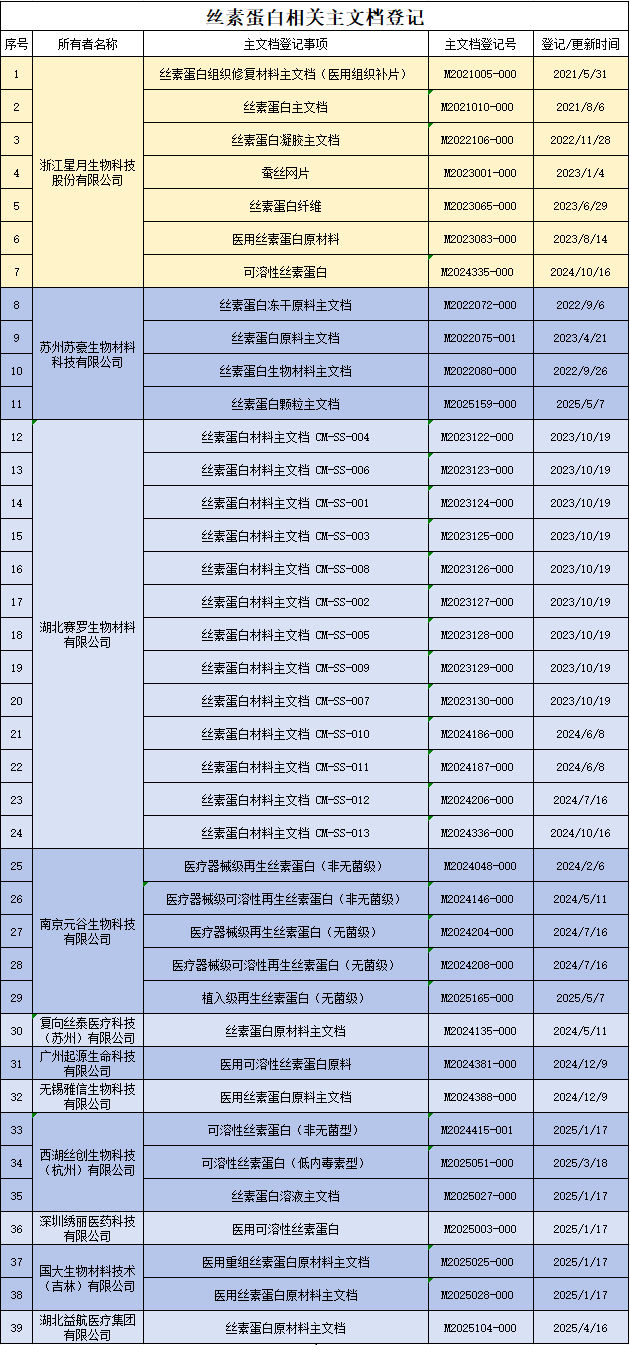
Information source: CMED website main document registration information disclosure
04 Practice of registering the main document of Star Moon Biology
Xingyue Biotechnology has been deeply involved in the field of silk materials for 20 years. Through continuous innovation, the company has acquired core technologies such as high-purity silk fibroin extraction, molecular recombination, and enzyme degradation. It has successfully prepared silk fibroin raw materials in various forms such as fibers, powders, solutions, and sponges. Since the implementation of the main document system by the National Medical Products Administration, Xingyue Biotechnology has taken the lead in carrying out the registration of silk fibroin material main documents in the industry, promoting industry compliance development through systematic technical filing.
In 2021, Xingyue Biotechnology completed the registration of the main document for silk fibroin tissue repair materials (medical tissue patches), officially embarking on a new journey of registering the main document for silk fibroin raw materials. Up to now, the company has completed the registration of 7 main documents of silk fibroin core materials, including silk fibroin gel, silk mesh, silk fibroin fiber, soluble silk fibroin and other raw material types. These raw materials can be widely used in cutting-edge medical fields such as cell culture, tissue engineering, and regenerative medicine, providing traceable, safe, and innovative raw material solutions for the medical device market.

05 Triple empowerment of the main document system
The medical device master document system, as an innovative regulatory mechanism, has significantly optimized the medical device registration process by establishing a standardized technical data management system. While ensuring the security of core intellectual property rights of enterprises, it has brought comprehensive improvement to the medical device industry.
From the perspective of enterprises, it significantly reduces the cost and time consumption of research and development registration, enabling enterprises to invest more resources in core technological innovation; From the perspective of the industrial chain, it has promoted efficient collaboration between upstream and downstream enterprises, reduced redundant verification and audit processes, significantly shortened product registration cycles, and accelerated the product launch process; From the perspective of industry ecology, it has promoted the unification of technical standards and the improvement of regulatory efficiency, guiding the industry to transform from low-end imitation to high value-added innovation. Overall, the medical device master document system not only optimizes the registration process for individual products, but also injects new momentum into the high-quality development of the entire industry through the establishment of a sustainable technology sharing mechanism, achieving multiple goals of intellectual property protection, industrial efficiency improvement, and innovation ecosystem cultivation.









