Good news | Zhejiang Xingyue Biotechnology Co., Ltd. has obtained the National Class II Medical Device Product Registration Certificate

Recently, the Class II medical device "silk protein hydrogel dressing", which was independently developed by Zhejiang Xingyue Biotechnology Co., Ltd. with mulberry silk as raw material, was officially granted the Registration Certificate of Medical Device Products (Registration Certificate No.: [ZXZZ 20222140466]) by Zhejiang Food and Drug Administration.
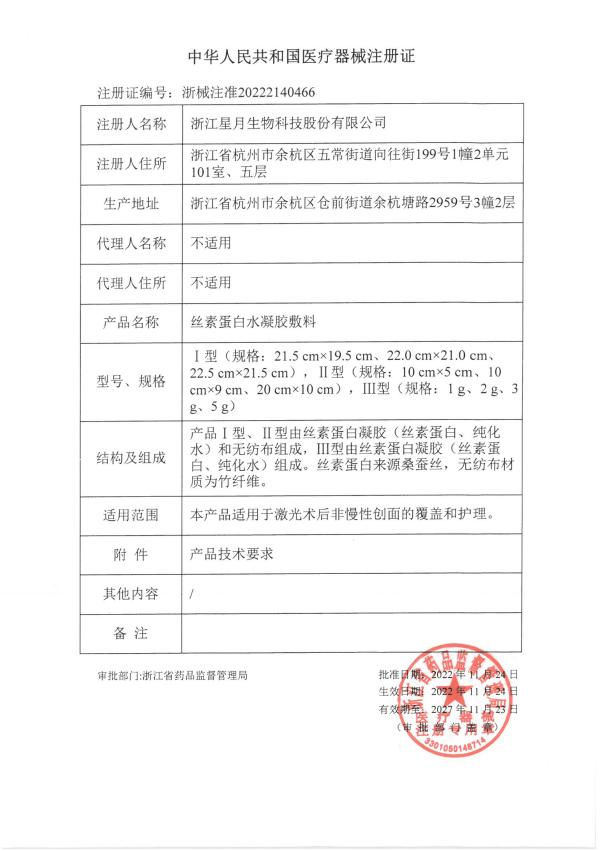
The product has three types: type I, type II and type III, of which type I and type II are composed of silk fibroin gel (silk fibroin protein, purified water) and non-woven fabric, and type III is composed of silk fibroin gel (silk fibroin protein, purified water). The silk fibroin used is derived from mulberry silk, and the non-woven fabric material is bamboo fiber.
The product is mainly suitable for covering and caring for non chronic wounds after laser surgery, with good liquid affinity and continuous supply of water to the wound.
The successful approval of the company's medical device registration certificate means that the hydrogel series products independently developed by Xingyue Biology have been approved for marketing, marking another solid step in the medical field of "silk fibroin". This not only reflects the company's strong research and development capabilities, but also showcases the spirit of unity and courage among the employees of the biological company with the strong support of the group company. Our team is a cohesive, capable of fighting tough battles, and full of vitality!
In the future, the company will continue to increase research and development investment in the field of innovative medical devices, in order to achieve more breakthroughs.









